Introduction
The Centers for Medicare and Medicaid Services (CMS) has posted its proposed rule for the 2022 Medicare Physician Fee Schedule (PFS), including proposals for changes to the Quality Payment Program (QPP).1. The QPP, effective since 2017, has consisted of 2 tracks for participation by eligible Medicare Part B clinicians. Under QPP, providers could choose to participate through either of 2 value-based reimbursement pathways: the Merit-based Incentive Payment System (MIPS) or the advanced APMs.2 With close to 100% of MIPS-eligible clinicians engaged in these value-based quality payment programs,3 plans proposed by CMS to begin transitioning the traditional MIPS program to MIPS Value Pathways (MVPs) in 2023, and the signaled intent to move completely to digital quality measures (potentially as early as 2025),4 the Biden administration has indicated its intent to continue with value-based reimbursement models, with modifications based on “lessons learned.”
Proposed Changes: 2022
More imminent, however, are proposed changes that could be effective as early as next year, including changes to quality measures and improvement activities. The QPP has 4 performance categories potentially impacting reimbursement: quality, cost, interoperability, and improvement activities. Depending on the model clinicians participate in, performance on Quality will be weighted 30% to 55% (eg, 30% for traditional MIPS) in calendar year (CY) 2022, and improvement activities will be weighted 15% to 20% (eg, 15% for traditional MIPS).5 Stakeholders (pharmaceutical manufacturers, clinicians, health plans, and systems) should take note of the proposed changes in quality measures and improvement activities, identify the potential impacts (challenges and opportunities) upon their practices and relevant therapeutic areas, and formulate strategies and tactics to support access and alignment with the Quintuple Aim (Population Health, Care Team Well-Being, Patient Experience, Equity & Inclusion, Reducing Cost).6
Quality Measures: Mapping Next Steps
CMS is proposing 195 quality measures for the 2022 performance period,1,5 which include:
- Significant changes to 84 existing MIPS quality measures
- Changes to the specialty sets (more than 40 in 2021),7 including removal of select measures within specific sets, and addition of new outcome and intermediate outcome measures
- Addition of 2 new specialty groups
- Certified nurse midwife and corresponding new specialty measure set
- Clinical social work is also a proposed new MIPS-eligible clinician, with proposed specialty set changes
- Removal of 19 quality measures
- Addition of 5 quality measures, which include 2 new administrative claims measures, and request for public comment on the draft COVID-19 vaccination by clinicians measure specifications
The administrative claims measures being proposed reflect CMS’s continued concerns regarding hospital admission rates and are:
- Risk-Standardized Acute Unplanned Cardiovascular-Related Admission Rates for Patients with Heart Failure for the MIPS (applicable to MIPS eligible clinicians, groups, virtual groups)
- Clinician and Clinician Group Risk-standardized Hospital Admission Rates for Patients with Multiple Chronic Conditions (applicable to MIPS eligible groups with at least 16 clinicians)
It is recommended that manufacturers:
- Identify current and proposed new or modified quality measures in relevant data sets (eg, QPP, Health Effectiveness Data and Information Set [HEDIS], Medicare Stars Part C and Part D) and applicable to your therapeutic area
- Review the literature relevant to your portfolio for evidence of support of quality measure performance (eg, readmission reduction)
- Identify and address quality measure data gaps (eg, plan for real-world data collection and analyses)
- Develop a plan to communicate product performance supportive of relevant quality measures, including updating value message frameworks
- In addition, if quality metrics reflecting gaps in care are not yet available, identify opportunities for multistakeholder collaborations to formulate quality measure concepts intended to expedite closure of care gaps through evidence-based practices and therapies
PRECISIONvalue’s Quality Measure Analysis Map
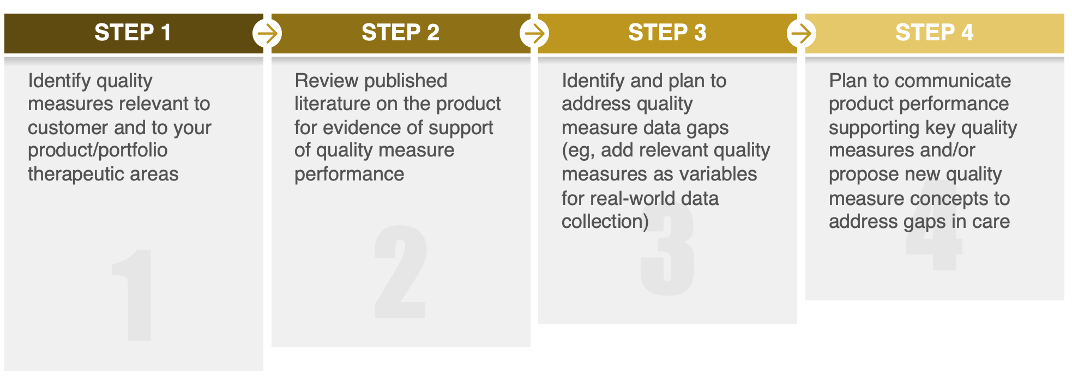
Additional information about PRECISIONvalue’s Quality Measure Analysis Road Map process is available through your PRECISIONvalue Account Manager.
Improvement Activities: Advancing to the Quintuple Aim

The National Academy of Medicine has been calling for “…a new Quintuple Aim of equity and inclusion for health and healthcare.”6 The Agency for Healthcare Research and Quality (AHRQ) has also provided examples of operational outcomes measures (eg, health disparities, social determinants of health [SDOH]) and metrics that could be used to measure progress toward attainment of this Fifth Aim.8 In their proposed rule for QPP 2022, CMS has proposed 7 new and 15 revised improvement activities (while deleting 6 previous ones). Many of these proposed new or revised activities help to address the Fifth Aim of Equity and Inclusion, and examples1 are:
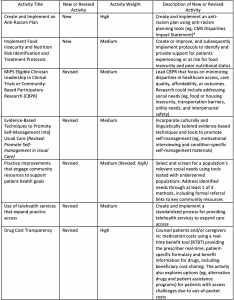
In a 2021 PRECISIONvalue survey of stakeholders (including health systems and ACOs), >80% of respondents were at least willing to consider future collaborations with manufacturers on an array of SDOH activities. Some stakeholders had manufacturer collaborations to address SDOH (eg, access to drugs or telehealth) underway. With nearly 100% of MIPS-eligible clinicians engaged with the program, opportunities exist to support organized clinicians with their QPP improvement activities. Pharmaceutical manufacturers should monitor the changes in improvement activities once the final rule is published later this year and conduct a landscape analysis to identify relevant activities for your therapeutic areas and customers. For more information on PRECISIONvalue’s resources to support these analyses, value-based alliances, and customers’ SDOH activities, please contact your PRECISIONvalue Account Manager.
References
- Department of Health and Human Services. Medicare Program; CY 2022 Payment Policies Under the Physician Fee Schedule and Other Changes to Part B Payment Policies; Medicare Shared Savings Program Requirements; Provider Enrollment Regulation Updates; Provider and Supplier Prepayment and Post-Payment Medical Review Requirements. https://public-inspection.federalregister.gov/2021-14973.pdf. Accessed October 14, 2021.
- McDermott+ Consulting. MACRA final rule released: CMS establishes 2016 as transition year for QPP. https://www.mcdermottplus.com/insights/macra-final-rule-released-cms-establishes-2017-as-transition-year-for-qpp/. Accessed October 14, 2021.
- Centers for Medicare & Medicaid Services. Quality Payment Program participation in 2019: results at-a-glance. https://qpp-cm-prod-content.s3.amazonaws.com/uploads/1190/QPP%202019%20Participation%20Results%20Infographic.pdf. Accessed October 14, 2021.
- MDinteractive. CMS releases 2022 MIPS proposed rule – key takeaways. https://mdinteractive.com/mips-blog/cms-releases-2022-mips-proposed-rule-key-takeaways. Published July 22, 2021. Accessed October 14, 2021.
- Centers for Medicare & Medicaid Services. QPP 2022 proposed rule fact sheet. In: 2022 Quality Payment Program proposed rule resources. https://qpp.cms.gov/resources/resource-library. Accessed October 6, 2021.
- Matheny M, Israni ST, Ahmed M, Whicher D, eds. Artificial intelligence in health care: the hope, the hype, the promise, the peril. National Academy of Medicine website. https://nam.edu/wp-content/uploads/2019/12/AI-in-Health-Care-PREPUB-FINAL.pdf. Published December 2019. Accessed October 14, 2021.
- Physicians Advocacy Institute. Frequently asked questions. http://www.physiciansadvocacyinstitute.org/MACRA-QPP-Center/FAQs/mips-quality-category-4-what-are-specialty-measure-sets. Accessed October 14, 2021.
- Coleman K, Wagner E, Schaefer J, Reid R, LeRoy I. Redefining primary care for the 21st century: white paper. AHRQ Publication No. 16(17)-0022-EF. Rockville, MD: Agency for Healthcare Research and Quality; October 2016. https://www.ahrq.gov/ncepcr/tools/workforce-financing/white-paper.html#tab2. Accessed October 14, 2021.
- Centers for Medicare & Medicaid Services. Disparities impact statement. https://www.cms.gov/About-CMS/Agency-Information/OMH/Downloads/Disparities-Impact-Statement-508-rev102018.pdf. Revised March 2021. Accessed October 14, 2021.


